Ancar Sd-150 Manuel utilisateur

Sd-150
USER MANUAL
Rev.2 July 2020


USER MANUAL DENTAL UNIT SD-150 - REV. 2 JULY
1
PAGE
1. OVERVIEW 2
2. WARRANTY 2
3. IDENTIFICATION 4
4. PRECAUTIONS 4
5. CLAUSES 6
6. TECHNICAL FEATURES 7
6.1. Chair 7
6.2. Dental Unit 7
7. TRANSPORT AND DIMENSIONS 8
8. DESCRIPTION OF THE DENTAL UNIT 9
8.1. Connections box 10
8.2. Pneumatic pedal 11
8.3. Instrument tray 12
8.4. Main keypad 14
8.5. Hydric unit 15
8.6. Hose support 17
8.7. Auxiliary keypad 18
8.8. Headrest 19
8.9. Optional armrest 19
9. SAFE MOVEMENTS 20
10. CLEANING AND STERILISATION 20
11. TECHNICAL DATA 21
ANNEX 1.
CLEANING AND DISINFECTION OF THE CATTANI TANK, HOSES AND BASIN
22
ANNEX 2. VS300 SUCTION VALVES KIT (DÜRR DENTAL) 23
ANNEX 3. TYP1 SUCTION SYSTEM (METASYS) 24
ANNEX 4. ECO SUCTION SYSTEM (METASYS) 25
ANNEX 5. SELF WATER SYSTEM KIT 26
ANNEX 6. CAS 1 COMBI-SEPARATOR (DÜRR DENTAL) 27
ANNEX 7. CS 1 SEPARATOR (DÜRR DENTAL) 27
ANNEX 8. WEK WATER DECONTAMINATION SYSTEM: (METASYS) 28
ANNEX 9. H1 HYGIENE SYSTEM (METASYS) 29
ANNEX 10. MINILIGHT SYRINGE (LUZZANI) 30
ANNEX 11. OPTIONAL COMPONENTS 33
ANNEX 12. CLEANING AND DISINFECTION OF OTHER EQUIPMENT PARTS 34
ANNEX 13. SAFETY OBSERVATIONS 35
ANNEX 14. SYMBOL DEFINITIONS 37
CONTENTS

USER MANUALDENTAL UNIT SD-150 - REV.2 JULY 2020
2
1. OVERVIEW
Thank you for purchasing the SD-150 dental unit.
This instruction manual contains information about the dental device, its set-up and maintenance.
Notes
This symbol means ALERT, CAUTION.
The User Manual must be read and understood before starting up the unit.
This manual should be kept in a safe place for future reference, for as long as
you use the equipment.
Follow all safety standards.
The user is in charge of keeping the unit in perfect operating conditions, clean
and disinfected.
This dental equipment is intended to be used exclusively by a professional user who must be
properly trained and licensed to practice dental medicine.
This equipment can only be handled by an authorised technical service.
Keep the unit installed in an environment under controlled conditions, including temperature
range (+10˚C to +40˚C), humidity (30-75%) and atmospheric pressure (700 to 1060 hPa), free
of condensation and dust and not exposed to direct sunlight.
The electrical installation of the premises where the equipment is installed must comply with the
instructions set forth in standard EN 60601-1 referring to electrical protections against electric
shock for Class I equipment.
Antoni Carles, S.A. reserves the right to make improvements or changes to this dental
equipment without prior notice.
The equipment must be used according to the instructions of use.
As per Directive 93/42/EEC, the dental unit and orthordintic chair manufactured by Antoni
Carles, S.A. are Class IIa equipment. It is strictly forbidden to install any Class IIb or Class III
dental instruments, such as surgical lasers, electro scalpels, X-rays, electrical cauterizers. Only
Class I or Class IIa devices can be installed and these must comply with the requirements set
forth in the aforementioned directive and harmonised standards EN60601-1 and EN60601-1-2.
2. WARRANTY
The units manufactured by ANTONI CARLES, S.A. are guaranteed against material and
manufacturing defects for 2 years from the date of installation, which must be registered
correctly in an online form. During this period, ANTONI CARLES, S.A. assumes the repair of
those parts or components of the unit that, once veried, are recognised as defective.
Under no circumstances shall ANTONI CARLES, S.A. be held responsible for defects and
damage caused by:
Natural wear of parts and components such as seals, lters, etc.
Inappropriate use, maintenance or cleaning of the unit and its components.
Accidental impacts or any other accidental cause or effects caused by user negligence.
Causes unrelated to the design or manufacture of the product or resulting from natural
phenomena such as electrical power outages, oods, etc.
Defects, intended damage or modications caused by third parties unrelated to Ancar.
Non-compliance with the maintenance periods specied in the User Manual or resulting from
inadequate repair or maintenance that has not been carried out by an ofcial Ancar technical
service.

USER MANUAL DENTAL UNIT SD-150 - REV. 2 JULY
3
The use of spare parts that are not original, recommended or manufactured by Ancar.
ANCAR shall not be held responsible under any circumstances for damage and losses derived
from defective parts or materials, whether direct or consequential.
In no case shall ANCAR be required to replace the entire unit or to reimburse its purchase price.
ANTONI CARLES, S.A.'s responsibility and accountability for any damages shall cease in the
following cases, whereby the warranty shall be automatically cancelled:
1. If the registry is not formalised by the distributor within 30 days from the date of installation.
2.
If interventions on the device are not carried out by technicians certied by ANTONI CARLES, S.A.
3. If incorrect maintenance or installation operations are carried out on the unit, or non-original
spare parts are used.
4. If the installations required for operation of the unit (electrical, water and pneumatic) are
carried out without observing the instructions in the Installation Manual or the provisions of the
current standards of the country of use are not observed.
5. If non-authorised modications are made on the unit, or other medical devices and/or
accessories not foreseen by ANTONI CARLES S.A. are connected to it.
6. If the maintenance schedule included in the User Manual is not followed.
Ancar does not cover any labour costs for repairs under the warranty.
The warranty for the integrated elements of other brands, will be that granted by each of the
respective manufacturers, and for which ANTONI CARLES S.A. shall not assume any liability
or obligation with regard to said warranty.
To obtain technical service, please contact an ofcial distributor of ANTONI CARLES,S.A.
(refer to the list of distributors in:
- SPA: www.ancar-online.com/distribuidores-dentales/
- ENG: www.ancar-online.com/en/distribuidores/
- FR: www.ancar-online.com/fr/distributeurs/
- DE: www.ancar-online.com/de/fachhaendler/).
ANTONI CARLES, S.A. reserves the right to modify the warranty conditions available for the
Customer at any time, insofar as permitted by current legislation.

USER MANUALDENTAL UNIT SD-150 - REV.2 JULY 2020
4
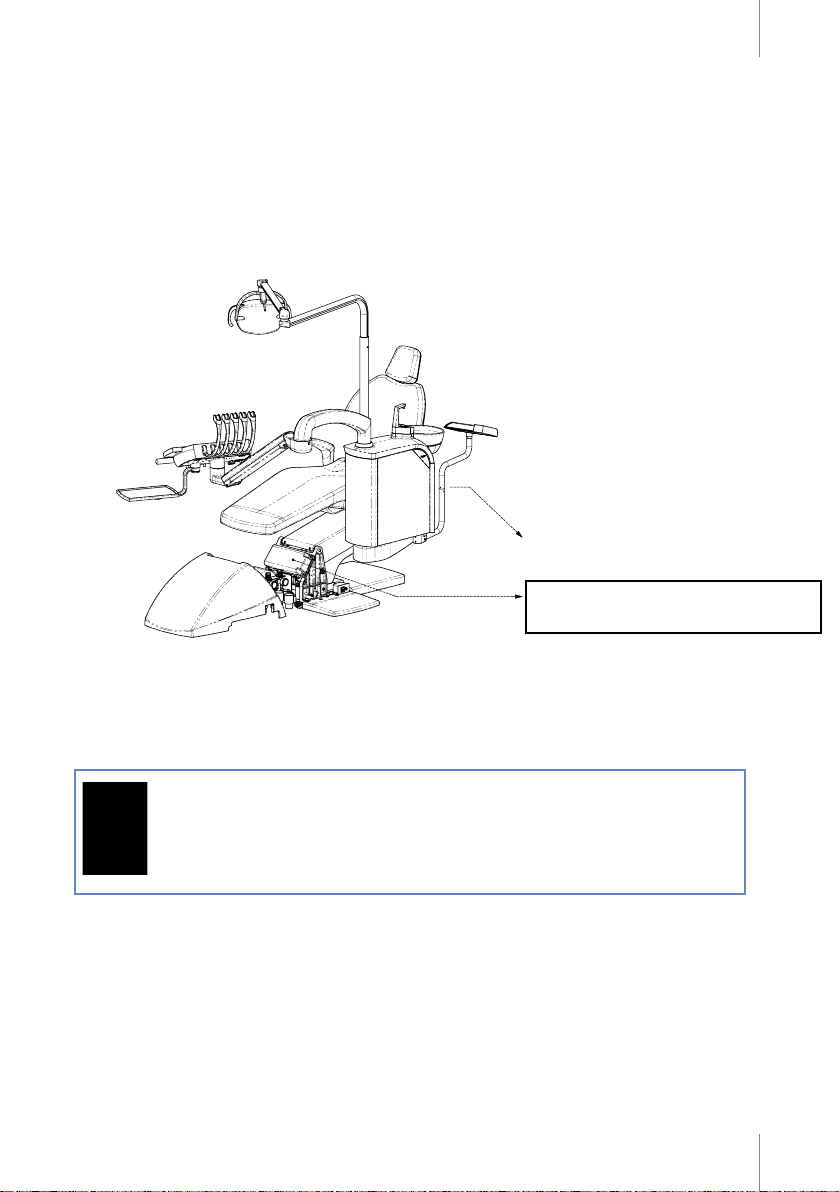
3. IDENTIFICATION
The identication label, which contains the technical information needed for connection, is
folded.
It can be seen from the rear of the seat and is also placed near the base, on the side of the chair.
This second label will be seen once the outer cover is removed (Fig. 1).
4. PRECAUTIONS
The unit must be installed in a location with plenty of light, away from areas transitted by people
and must have enough free space available to ensure that the patient and professional are
comfortable.
The dental chair must be anchored to the oor to guarantee stability.
Before moving the chair, the operator (doctor, user) must ensure that nobody else (patient,
companion, children) is in the chair's eld of action. Be especially careful as regards persons
present in the area of the backrest and the instrument tray.
After using the equipment, disconnect all the instruments and the unit. The unit must be
switched off at the end of the work day.
If the unit is not going to be used for a long period of time, the plug to the power supply must also
be disconnected and the main water and air valves should be closed.
Make sure that the main switch (Fig. 4, "J") is disconnected if the equipment is not under
direct supervision by staff. This will prevent the water intake ducts from withstanding constant
pressure. To prevent equipment malfunctions and damage to the property, the air and water
intake pressures must not exceed 6 bar and the hoses must not be exposed to temperatures
over 46ºC.
Do not overload the unit to maintain the useful life of the lifting unit components.
It is not advisable to use mobile phones near the unit when it is in operation.
This equipment is not designed to be used inside surgery rooms.
USER MANUAL DENTAL UNIT SD-150 - REV. 2 JULY
5
PRESERVATION OF THE ENVIRONMENT
All the materials used for packaging respect the environment and are recyclable: wooden
pallet, cardboard, polyethylene bag and bubble lm. Collection of the materials used favours
recovery, recycling and reduction of waste material.
Antoni Carles, S.A. is committed to achieving the objectives set forth in European Community
Directives 2011/65/EC and 2012/19/EC.
This symbol is only applicable for member states of the European Union.
To prevent potential negative consequences for the environment or human
health, this equipment must be disposed of (i) in EU member countries -
according to the WEEE (Directive on Waste Electric and Electronic Equipment)
and (ii) in all other countries, according to local regulations and recycling
legislation.
EMC
The dental equipment model Sd-150 complies with the essential requirements that apply to it
as set forth in the Medical Device Directive 93/42/EEC and meets the design and construction
requirements contained in standard EN60601-1-2 on the Safety of Medical Electrical Equipment
and Electromagnetic Compatibility; therefore it does not cause electromagnetic disturbances
and complies with immunity standards.
SN: N (Electro-pneumatic unit)+number
Nxxxx
Location of the label

USER MANUALDENTAL UNIT SD-150 - REV.2 JULY 2020
6
ENVIRONMENTAL CONDITIONS
When packaged for transport and storage, the equipment can withstand exposure up to 15
weeks to environmental conditions that do not exceed:
a) ambient temperatures between -20˚C and +50˚C.
b) relative humidity between 10% and 100%, including condensation.
c) atmospheric pressure between 500 hPa and 1060 hPa (from 500 mbar to 1060 mbar).
- Operating conditions:
d) ambient temperatures between +10˚C and +40˚C.
e) relative humidity between 30% and 75%, including condensation.
f) atmospheric pressure between 700 hPa and 1060 hPa (from 700 mbar to 1060 mbar).
5. CLAUSES
Antoni Carles, S.A. shall not be liable for damage due to re, natural disasters, actions
undertaken by third parties or other accidents (caused by the negligence or misuse of the
operator) or due to the use of the unit under unusual conditions.
Antoni Carles, S.A. shall not be liable for damage derived from inappropriate use of the device,
such as loss of business or loss of prots.
Antoni Carles, S.A. shall not be liable for the results of diagnoses made by a doctor when
using this device.

USER MANUAL DENTAL UNIT SD-150 - REV. 2 JULY
7
6. TECHNICAL FEATURES
6.1. Chair
Ergonomic design for the patient.
Chair movements powered by lifting gears that are silent and fully reliable.
Movements controlled by a microprocessor:
- Automatic return to zero movement (adjustable height).
- Automatic return movement to the cuspidor.
- 3 free positioning memories (settings for seat height and backrest).
High-quality and hygienic anatomic upholstery.
Safe backrest and seat movements, with automatic unlocking manoeuvre.
Adjustable headrest height and positioning.
Trendelenburg position included.
6.2. Dental Unit
Easy care and maintenance paint.
Instrument tray for 5 instruments and control panel with 14 functions. Includes syringe.
Optional: adjustable electric micromotor. Reversible direction of rotation.
Connection available for instruments equipped with lighting (micromotor, turbine, ultrasound).
Hydric box with 70º rotation, cuspidor with automatic cup ller and basin cleaning. A variety of
suction and amalgam systems can be ordered separately.
Arm movements compensated with a pneumatic brake, with an auxiliary instrument tray.
Connections box and tubes located at the front of the chair.
Control panels on the instrument tray and hose support.
The equipment has been designed and constructed so that no tubes or cables are visible from
the outside, with the exception of the hoses on the instrument tray.
The equipment complies with the essential requirements that apply to it, as set forth in the
Medical Device Directive 93/42/EEC, meets the design and construction requirements contained
in standards EN60601-1 and EN60601-1-2 on the Safety of Medical Electrical Equipment and
Electromagnetic Compatibility and has undergone a Risk Management analysis in accordance
with standard ISO 14971.
USER MANUALDENTAL UNIT SD-150 - REV.2 JULY 2020
8
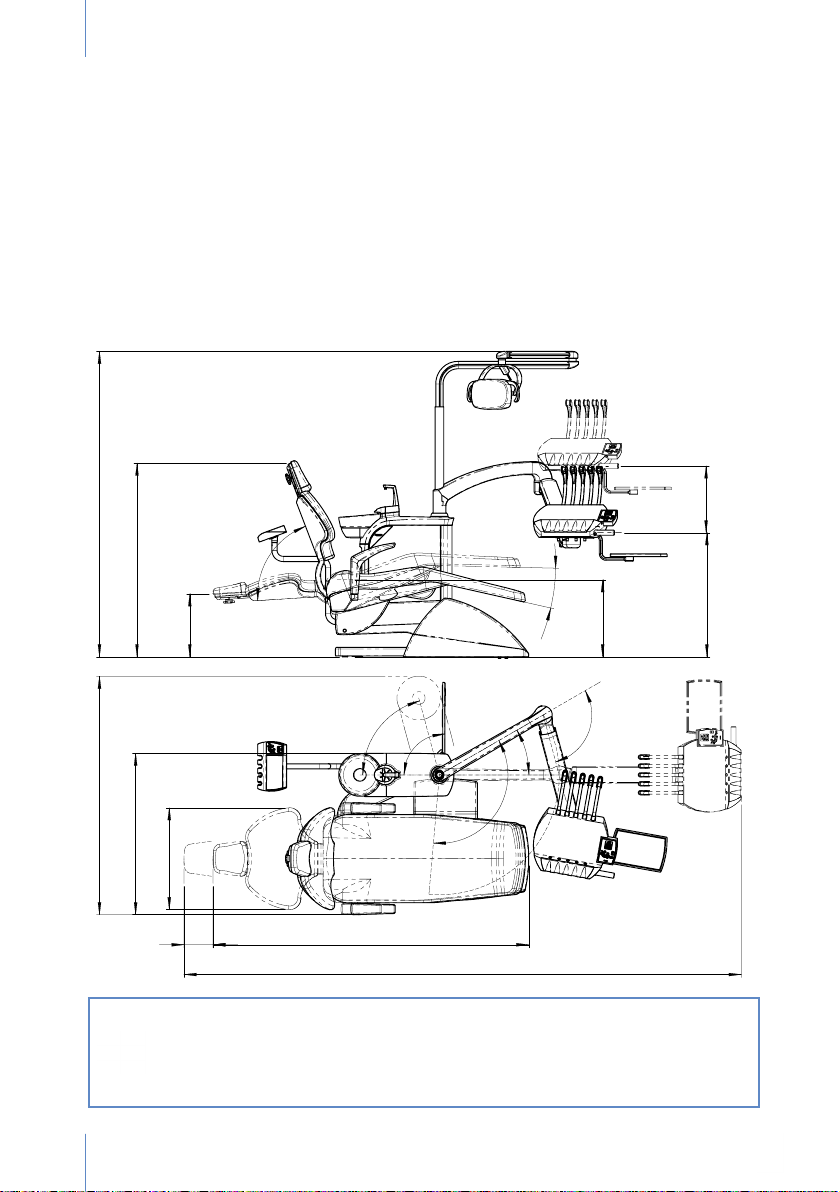
7. TRANSPORT AND DIMENSIONS
The dental unit is delivered perfectly packaged and protected.
One box contains the dental chair attached to a pallet and the other two boxes contain the
hydric box and whip arm system, respectively. (Figure 2)
It is essential that none of these boxes undergo impacts during transport and they must not fall on
the ground under any circumstance. We urge the utmost caution to be exercised during transport
of the equipment, which must be carried out by technical staff authorised by Antoni Carles, S.A.
Before the equipment is assembled, for your convenience and to ensure ergonomic working
conditions, a technician will accompany you and any authorised staff at the work site to plan
the ideal location for the equipment.
If the unit has to be transported after being assembled, position the seat of the
chair at its minimum height with the backrest raised and make sure the hydric
box and instrument tray unit is placed and secured as far back as possible on
the chair at all times.
Remember to anchor the chair to the oor at its new location.
0$;
0,10$;
0$;
0,1
0$;
0,1
0$;
0,1
0$;
Fig. 2
Autres manuels pour Sd-150
1
Table des matières
Autres manuels Ancar Équipement dentaire
Manuels Équipement dentaire populaires d'autres marques

Vatech
Vatech EzRay Air VEX-P300 Manuel utilisateur

KaVo
KaVo GENTLEpower LUX Contra-angle 25 LP Manuel utilisateur

DENTSPLY
DENTSPLY SmartLite Focus Manuel utilisateur

LM
LM ProPower CombiLED Manuel utilisateur

Owandy Radiology
Owandy Radiology RX-AC Manuel utilisateur
mectron
mectron Piezosurgery Manuel utilisateur


















